
Age-Defying Retinol%2, Peptide Serum, Immediate Effect Formula, Satisfactory Result in Short Time, Anti-Wrinkle, Hydrating,Plumping, Anti Aging, Dark Spot Corrector, Premium Skin Care Serie
Product details
| Management number | 213990906 | Release Date | 2026/04/12 | List Price | $16.00 | Model Number | 213990906 | ||
|---|---|---|---|---|---|---|---|---|---|
| Category | |||||||||
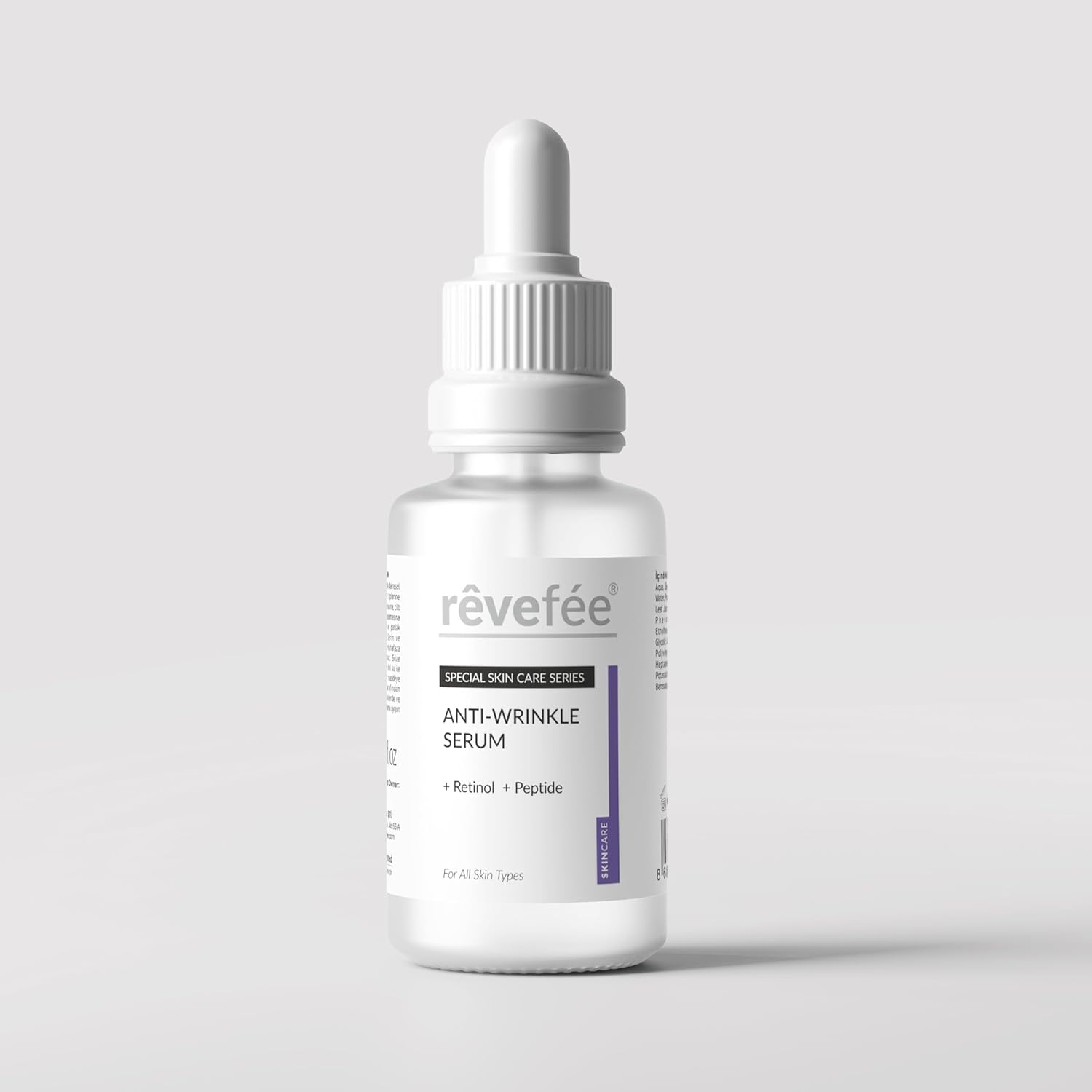
Transform your skincare routine with revefee's Anti-Wrinkle and Anti-Aging Skin Care Serum. This powerful formula combines the proven benefits of Retinol, Peptides, and Glycerin to help revitalize your skin. The carefully balanced blend works to address multiple skin concerns, targeting the appearance of wrinkles while supporting your skin's natural renewal process. The serum's versatile formula helps reduce the appearance of puffiness and supports overall skin health. Perfect for those seeking a comprehensive skincare solution, this serum is designed to complement your daily beauty regimen. The lightweight texture absorbs quickly, making it suitable for various skin types. For best results, incorporate this serum into your evening skincare routine and follow with your regular moisturizer. Experience the transformative effects of this premium skincare solution that brings together science-backed ingredients in one elegant formula.
- ADVANCED FORMULA: Premium skincare serum featuring a powerful combination of Retinol and Peptides for comprehensive anti-aging benefits. Immediate effect of reducing wrinkles.Combine the rejuvenating powers of retinol with the deep hydration of hyaluronic acid, effectively reducing the appearance of fine lines, wrinkles, and dark spots.
- ANTI-WRINKLE POWER: Specialized formula helps reduce the appearance of fine lines and wrinkles while promoting skin renewal.
- PUFFINESS REDUCTION: Targeted ingredients work to minimize under-eye puffiness and promote a more refreshed appearance.
- SKIN REVITALIZATION: Enhanced with Retinol to help maintain skin moisture while improving overall skin texture and appearance.This formula delivers necessary hydration, soothing relief, comprehensive care & gentleness for both dry & sensitive skin
- GENTLE CARE: Carefully formulated to be suitable for sensitive skin types while delivering effective anti-aging results.
- PREMIUM SKIN CARE: Quick effect of reducing wrinkles. Satisfactory result in short time. The serum's versatile formula helps reduce the appearance of puffiness and supports overall skin health.
| Item Form | Serum |
|---|---|
| Skin Tone | All |
| Skin Type | All |
| Brand Name | rêvefèe |
| Scent Name | Rosa Damascena |
| Unit Count | 1.01 Fluid Ounces |
| Item Volume | 1.01 Fluid Ounces |
| Item Weight | 89 Grams |
| Manufacturer | TULCA GROUP DIS TICARET SANAYI TICARET LIMITED SIRKETI |
| Number of Items | 1 |
| Product Benefits | Skin revitalization, Anti-puffiness, Wrinkle reduction |
| Material Features | Anti-aging formula, Retinol+Peptide Premium Skin Care Formula |
| Active Ingredients | Retinol, Peptide, Hyaluronic Acid, Hyaluronate,Glyserin, Glycolic Acid, glycolic_acid, lactic_acid |
| Material Type Free | Paraben Free |
| Special Ingredients | Glycolic Acid, Hyaluronate, Peptide, Retinol |
| Target Use Body Part | Face |
| Recommended Uses For Product | Anti-wrinkle, Anti-aging, Skin revitalization, fine lines, dark spot corrector |
Correction of product information
If you notice any omissions or errors in the product information on this page, please use the correction request form below.
Correction Request Form







![[Serment] Korean Cosmetics Serment Peptide & Stem Cell Serum 30ML Korea Premium Brand ADVANCED SUPER LIPOSOME](https://m.media-amazon.com/images/I/512I6dzf1LL._SL1000_.jpg)










